Estimated Read Time: 20 minutes | Target Exam: USMLE Step 1 | Difficulty: Beginner to Advanced
Why Pathology Is the Backbone of USMLE Step 1
If pharmacology is the muscle of USMLE Step 1, pathology is the skeleton. It underlies nearly every question on the exam, regardless of which organ system is being tested.
Here’s what makes pathology uniquely important:
The NBME content outline lists pathology as contributing to approximately 44–56% of Step 1 questions when you include pathophysiology. That means you can’t “skip” or “minimize” pathology and still score competitively. Whether the question is about a cardiovascular drug, an infectious disease, or a genetic disorder — it’s almost always grounded in a pathological process.
The students who struggle most with Step 1 are the ones who treat pathology like a memorization subject. They try to memorize buzzwords (“apple-green birefringence = amyloid”) without understanding why amyloid behaves that way, or how that connects to the clinical presentation. That’s backwards.
This guide gives you real USMLE-style pathology practice questions for Step 1 with deep, reasoning-based explanations — plus the high-yield concepts, study strategies, and pattern recognition skills that will help you turn pathology from your biggest weakness into your most reliable score source.
What Makes a Great USMLE Pathology Question?
Before diving into practice, you need to understand how NBME pathology questions are constructed. The NBME never asks:
“What is the definition of dystrophic calcification?”
Instead, they give you a clinical scenario + histology description + lab findings and ask you to:
- Identify the underlying pathological mechanism
- Connect the pathology to a specific disease or condition
- Predict the clinical consequence of a pathological finding
- Distinguish between two closely related pathological processes
The golden rule: Every USMLE pathology question is asking you to connect mechanism → morphology → clinical presentation. Master that triangle and no question will catch you off guard.
High-Yield Pathology Categories for USMLE Step 1
Based on NBME content specifications and analysis of high-frequency testing areas, here are the categories you must own:
- Cell injury and death (necrosis types, apoptosis, free radicals)
- Inflammation and repair (acute vs. chronic, granulomatous disease, wound healing)
- Neoplasia (tumor characteristics, grading vs. staging, oncogenes, tumor suppressors)
- Genetic and developmental disorders
- Vascular pathology (atherosclerosis, thrombus vs. embolus, infarction)
- Immunopathology (hypersensitivity reactions, autoimmune diseases)
- Organ-specific pathology (cardiac, pulmonary, renal, hepatic, CNS)
- Hematopathology (anemias, leukemias, lymphomas)
- Infectious pathology (histological appearance of pathogens)
Now let’s work through targeted, high-quality practice questions.
usmle pharmacology questions with detailed explanation
USMLE Step 1 Pathology Practice Questions with Detailed Explanations
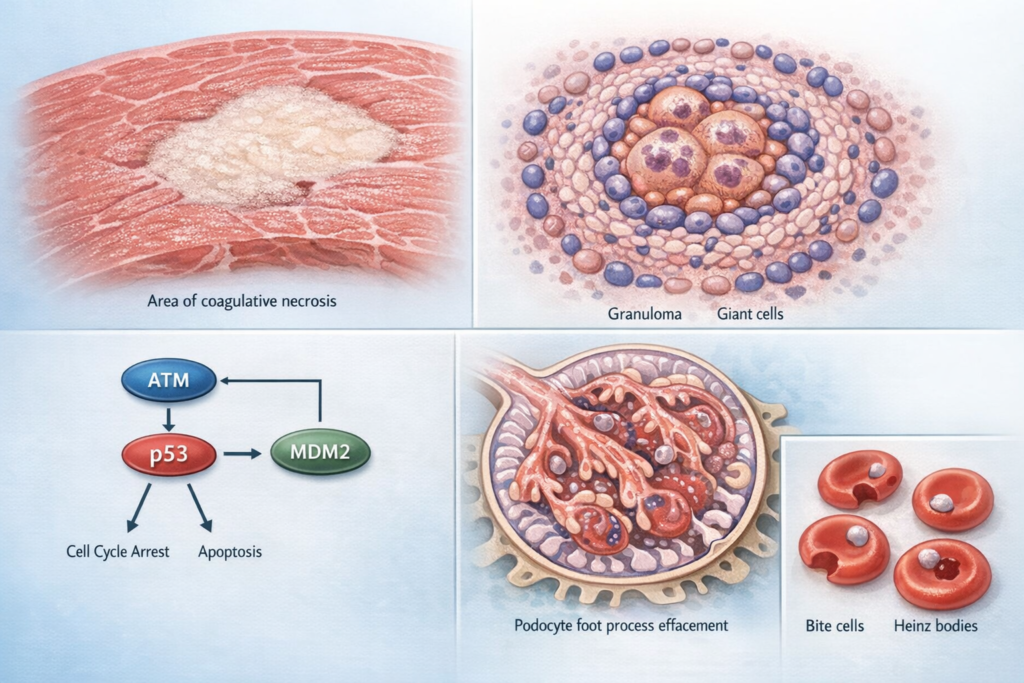
🔴 QUESTION 1 — Cell Injury & Necrosis (Foundational)
A 58-year-old man with a history of hypertension and hyperlipidemia presents to the emergency department with sudden-onset chest pain radiating to his left arm. He is found to have an ST-elevation myocardial infarction. He dies 3 days later despite maximal therapy. At autopsy, the myocardium in the affected region appears pale and firm, with preserved cellular architecture on histology but loss of nuclei. Which type of necrosis is this?
A) Liquefactive necrosis
B) Caseous necrosis
C) Fat necrosis
D) Coagulative necrosis
E) Fibrinoid necrosis
✅ Correct Answer: D — Coagulative necrosis
Detailed Explanation:
This is one of the most fundamental pathology questions on Step 1, and the NBME returns to it repeatedly in different clinical disguises.
Coagulative necrosis is the hallmark of ischemic injury in most solid organs (heart, kidney, liver, spleen — everywhere except the brain). The key histological feature is:
Preserved cellular architecture (tissue outline intact) + loss of nuclear staining (pyknosis → karyorrhexis → karyolysis)
Here’s the mechanism: When blood supply is cut off, the cell’s protein-synthesizing machinery fails. However, the structural proteins that form the cell’s scaffolding (cytokeratin, collagen) are denatured but not dissolved by the ischemic process. This is why the tissue looks like a “ghost” — cell outlines remain, but there’s no nuclear material (no living cells).
Macroscopic appearance: Pale, firm, wedge-shaped (in kidneys) or segmental (in heart). By 24–72 hours, neutrophils infiltrate the area — this is also why troponin rises and why histology at day 1–3 shows neutrophilic infiltration.
Why each distractor is wrong:
| Necrosis Type | Classic Location/Cause | Key Feature |
|---|---|---|
| Liquefactive | Brain ischemia, bacterial abscess | Tissue is digested to liquid pus; no preserved architecture |
| Caseous | TB, fungal infections | “Cheese-like” — amorphous pink acellular debris, surrounded by granuloma |
| Fat | Pancreatic lipase release (pancreatitis), breast trauma | Chalky white deposits (calcium soaps); saponification |
| Coagulative | Ischemia of solid organs (except brain) | Ghost cells; preserved architecture, no nuclei |
| Fibrinoid | Vasculitis, malignant hypertension, immune complex deposition | Vessels appear “smeared” with pink fibrin-like material |
The brain exception: Brain infarcts cause liquefactive necrosis (not coagulative) because the brain is rich in lipids and proteolytic enzymes — the tissue liquefies rather than coagulating.
USMLE Pattern to Recognize: Whenever you see ischemia + heart/kidney/spleen → coagulative necrosis. Whenever you see ischemia + brain → liquefactive.
🔴 QUESTION 2 — Inflammation & Granulomas (High-Yield)
A 34-year-old African American woman presents with progressive shortness of breath, bilateral hilar lymphadenopathy on chest X-ray, and elevated serum ACE levels. A transbronchial biopsy is performed. Histology shows collections of epithelioid macrophages and multinucleated giant cells surrounded by a rim of lymphocytes, without central necrosis. What is the most likely diagnosis?
A) Tuberculosis
B) Sarcoidosis
C) Histoplasmosis
D) Crohn’s disease
E) Cat scratch disease
✅ Correct Answer: B — Sarcoidosis
Detailed Explanation:
This question tests your ability to distinguish between granulomatous diseases — one of the highest-yield pathology concepts on Step 1.
What is a granuloma? A granuloma is a focal collection of activated macrophages (epithelioid macrophages) + multinucleated giant cells, representing a chronic inflammatory response to something the immune system cannot eliminate. Think of it as the body’s way of “walling off” a persistent threat.
The key distinguishing feature here is: NO central necrosis
This immediately rules out tuberculosis, which produces caseating granulomas (central caseous/cheese-like necrosis). The absence of necrosis, combined with the clinical picture (young African American woman, bilateral hilar adenopathy, elevated ACE), points unmistakably to sarcoidosis.
Sarcoidosis Pathology Pearls:
- Non-caseating (non-necrotizing) granulomas — the pathological hallmark
- Elevated ACE (produced by epithelioid macrophages in granulomas)
- Elevated serum calcium (macrophages hydroxylate vitamin D to its active form → hypercalcemia)
- Asteroid bodies (star-shaped inclusions in giant cells) and Schaumann bodies (laminated calcium inclusions) — buzzwords on histology
- Affects lungs, skin, eyes (uveitis), heart (arrhythmias), and nervous system
Granuloma Comparison Table — Memorize This:
| Disease | Caseating? | Key Features |
|---|---|---|
| Tuberculosis | ✅ Yes (caseating) | AFB+ on Ziehl-Neelsen stain; PPD+ |
| Sarcoidosis | ❌ No (non-caseating) | Elevated ACE, hypercalcemia, bilateral hilar LAD |
| Histoplasmosis | ✅ Yes (in immunocompromised) | Intracellular organism within macrophages; Ohio/Mississippi River Valley |
| Crohn’s disease | ❌ No | Transmural inflammation, skip lesions, GI tract |
| Cat scratch disease | ❌ No (stellate microabscesses) | Bartonella henselae, cat scratch, suppurative |
| Berylliosis | ❌ No | Occupational exposure; indistinguishable from sarcoid on biopsy |
| Leprosy | Variable | M. leprae; Virchow cells (lepra cells) |
USMLE Step 1 Glomerulonephritis Pathology Questions
🔴 QUESTION 3 — Neoplasia: Oncogenes vs. Tumor Suppressors (Step 1 Core)
A 45-year-old woman undergoes genetic testing after her mother and maternal aunt were both diagnosed with breast cancer before age 50. Testing reveals a mutation in a gene whose protein product normally phosphorylates and inactivates MDM2, thereby allowing p53 to remain active and trigger apoptosis in damaged cells. Loss of this gene is also associated with osteosarcomas. Which gene is most likely mutated?
A) BRCA1
B) RB1
C) TP53
D) TP53 upstream regulator (ATM)
E) HER2/neu
✅ Correct Answer: D — ATM (Ataxia-Telangiectasia Mutated)
Detailed Explanation:
This is a beautifully constructed Step 1 question designed to test whether you understand the p53 pathway in depth — not just that “p53 = tumor suppressor.”
The p53 Pathway: Normal cells suffer DNA damage → ATM kinase is activated → ATM phosphorylates MDM2 (inactivating it) → p53 is no longer degraded by MDM2 → p53 accumulates → p53 triggers cell cycle arrest (via p21) and apoptosis.
This patient has a mutation in the gene that sits upstream of p53: ATM.
Why ATM mutations cause cancer: Without functional ATM, MDM2 is never inactivated → p53 is constitutively degraded → cells can’t respond to DNA damage → mutations accumulate → cancer.
ATM mutations → Ataxia-Telangiectasia syndrome:
- Cerebellar ataxia (progressive)
- Telangiectasias (dilated blood vessels in eyes/skin)
- Immunodeficiency (IgA deficiency most common)
- Radiosensitivity (DNA damage can’t be repaired)
- Cancer predisposition: breast cancer (in female carriers), leukemia, lymphoma
Why the distractors are wrong:
- A (BRCA1): BRCA1 is involved in DNA repair via homologous recombination, not directly in the ATM-MDM2-p53 axis. Yes, associated with breast cancer, but the mechanism described doesn’t fit.
- B (RB1): The retinoblastoma gene; phosphorylated by CDK4/6 → releases E2F → cell cycle progression. A completely different pathway.
- C (TP53): p53 itself is the downstream effector here, not the upstream kinase described.
- E (HER2/neu): An oncogene (not tumor suppressor); tyrosine kinase receptor that promotes cell growth — amplified in 20–30% of breast cancers.
Oncogene vs. Tumor Suppressor — Core Concepts:
| Feature | Oncogene | Tumor Suppressor |
|---|---|---|
| Normal function | Promotes cell growth | Inhibits cell growth / promotes repair |
| Mutation type | Gain-of-function (dominant) | Loss-of-function (recessive — need 2 hits) |
| Analogy | Stuck accelerator | Broken brakes |
| Examples | RAS, MYC, HER2, BCL-2, cyclin D | TP53, RB1, BRCA1/2, APC, VHL, PTEN |
Knudson’s Two-Hit Hypothesis: For tumor suppressors, both alleles must be inactivated (“two hits”). In familial cancers, the first hit is inherited — so only one somatic mutation is needed to lose function (explains earlier onset in familial vs. sporadic cancers).
usmle step 1 practice questions with explanations free
🔴 QUESTION 4 — Vascular Pathology: Thrombus vs. Embolus (Clinical Integration)
A 72-year-old woman with a history of atrial fibrillation, poorly controlled, presents with sudden onset right-sided weakness and aphasia. CT scan of the brain shows a hypodense lesion in the territory of the left middle cerebral artery. Which of the following best describes the most likely origin and type of the pathological lesion?
A) In situ thrombosis of the left MCA due to atherosclerosis
B) Cardiogenic embolism from a mural thrombus in the left atrial appendage
C) Hypertensive small vessel disease causing lacunar infarct
D) Vasculitis-induced occlusion of the left MCA
E) Venous sinus thrombosis causing hemorrhagic infarction
✅ Correct Answer: B — Cardiogenic embolism from a mural thrombus in the left atrial appendage
Detailed Explanation:
This question tests vascular pathology, stroke mechanisms, and cardioembolic risk — all rolled into one vignette.
Why cardiogenic embolism:
Atrial fibrillation causes loss of coordinated atrial contraction → blood pools in the left atrial appendage (LAA) → stasis → thrombus formation (Virchow’s Triad: stasis, endothelial injury, hypercoagulability). These thrombi can embolize to cerebral vessels, causing ischemic stroke.
AF is the #1 cause of cardioembolic stroke in the elderly. The clot typically lodges in large vessels — the MCA territory is the most commonly affected because it receives ~80% of carotid blood flow.
Virchow’s Triad (Memorize This):
| Component | Examples |
|---|---|
| Stasis | Atrial fibrillation, prolonged immobility, heart failure |
| Endothelial injury | Atherosclerosis, hypertension, trauma, vasculitis |
| Hypercoagulability | Factor V Leiden, antiphospholipid syndrome, malignancy, pregnancy |
Why the distractors are wrong:
- A (In situ MCA thrombosis): This happens in atherosclerotic disease but typically produces a more gradual onset (“stuttering” neurological deficits) rather than sudden complete MCA territory infarction
- C (Lacunar infarct): These are small (< 1.5 cm), deep infarcts from small vessel disease due to hypertension or diabetes. They affect the basal ganglia, thalamus, internal capsule — not the entire MCA territory
- D (Vasculitis): Would present with signs of systemic inflammation and a different clinical picture
- E (Venous sinus thrombosis): Causes hemorrhagic infarction (not pale ischemic), often in younger patients or those with prothrombotic states; presents with headache, papilledema
Thrombus vs. Embolus at a Glance:
| Feature | Thrombus | Embolus |
|---|---|---|
| Definition | Clot formed in situ | Clot (or other material) that travels from elsewhere |
| Lines of Zahn | Present (alternating fibrin/platelet + RBC layers — arterial thrombi) | Absent |
| Common sources of embolism | LAA (Afib), LV mural thrombus (post-MI), DVT (→ PE), atheromatous plaque | — |
| Fat embolism | — | Long bone fractures, liposuction; classic triad: petechiae + hypoxemia + confusion |
| Air embolism | — | IV lines, neck wounds, decompression sickness; “mill wheel” murmur |
| Amniotic fluid embolism | — | Postpartum; DIC, respiratory distress |
🔴 QUESTION 5 — Immunopathology: Hypersensitivity Reactions (Very High-Yield)
A 24-year-old medical student receives a PPD (tuberculin skin test) for a hospital credentialing requirement. Forty-eight hours later, she develops a firm, raised, erythematous induration of 15 mm at the injection site. A biopsy of the lesion would most likely show which of the following?
A) IgE-mast cell degranulation with eosinophil infiltration
B) IgG and IgM antibody-mediated complement activation
C) Immune complex deposition in vessels with neutrophilic infiltration
D) CD4+ T lymphocytes and macrophages with perivascular infiltration
E) NK cell-mediated cytotoxicity
✅ Correct Answer: D — CD4+ T lymphocytes and macrophages with perivascular infiltration
Detailed Explanation:
The PPD (tuberculin) test is the textbook example of Type IV (Delayed-Type) Hypersensitivity — and the NBME tests this constantly, both directly and hidden within clinical scenarios.
Type IV Hypersensitivity — Key Features:
- Mediated by: T cells (CD4+ Th1 cells primarily), NOT antibodies
- Timing: 48–72 hours (hence “delayed” — the test is read at 48–72 hours)
- Mechanism: Prior sensitization to antigen → re-exposure → antigen-presenting cells activate memory CD4+ Th1 cells → Th1 cells secrete IFN-γ → recruits and activates macrophages → induration (firm swelling from macrophage/lymphocyte infiltration)
- Histology: Perivascular CD4+ T lymphocytes and macrophages — NO eosinophils, NO antibody deposition
The Four Hypersensitivity Types — The Highest-Yield Table in Pathology:
| Type | Mechanism | Timing | Examples | Key Mediator |
|---|---|---|---|---|
| Type I | IgE → mast cell degranulation | Minutes | Anaphylaxis, allergic asthma, urticaria, bee sting allergy | IgE, histamine |
| Type II | IgG/IgM → complement or ADCC | Hours | Hemolytic anemia, Goodpasture’s, myasthenia gravis, Graves’ | IgG/IgM, complement |
| Type III | Immune complex deposition | 4–10 hrs | Serum sickness, lupus, post-strep GN, Arthus reaction | Complement, neutrophils |
| Type IV | T cell mediated (no antibody) | 48–72 hrs | PPD test, contact dermatitis (poison ivy), organ rejection (chronic), MS, T1DM | CD4+ T cells, macrophages, IFN-γ |
Clinical Applications of Type IV:
- Contact dermatitis (poison ivy, nickel allergy) — same mechanism, different antigen
- Graft rejection (chronic) — CD4+ and CD8+ T cells destroy the transplanted organ over months/years
- Type 1 Diabetes — T cell destruction of pancreatic beta cells
- Multiple Sclerosis — T cells attack myelin
- Granuloma formation — persistent macrophage activation = granuloma (sarcoid, TB)
usmle step 2 ck practice questions with answers
🔴 QUESTION 6 — Hematopathology: Anemias (Step 1 Classic)
A 28-year-old African American man develops sudden-onset severe back and abdominal pain, dark urine, and jaundice 3 days after starting primaquine for malaria prophylaxis. Labs show: hemoglobin 8.2 g/dL (baseline 14.5), elevated LDH, elevated indirect bilirubin, decreased haptoglobin. Peripheral blood smear shows red blood cells with “bite cells” and “blister cells.” What is the pathophysiology?
A) Autoimmune destruction of RBCs by IgG antibodies
B) Osmotic fragility from loss of spectrin in RBC membrane
C) Decreased hemoglobin synthesis from iron deficiency
D) Oxidative damage to hemoglobin from inadequate glutathione reduction
E) Mechanical destruction of RBCs from fibrin strands in small vessels
✅ Correct Answer: D — Oxidative damage to hemoglobin from inadequate glutathione reduction
Detailed Explanation:
This is a G6PD deficiency question — one of the most commonly tested hemolytic anemias on Step 1, and for good reason: it integrates genetics, biochemistry, hematopathology, and clinical medicine perfectly.
The Biochemical Mechanism:
Glucose-6-phosphate dehydrogenase (G6PD) is the rate-limiting enzyme of the hexose monophosphate shunt (pentose phosphate pathway). Its job is to generate NADPH, which is required to reduce glutathione (oxidized → reduced form). Reduced glutathione is the RBC’s main antioxidant.
In G6PD deficiency: oxidative stress (from primaquine, dapsone, infection, fava beans) → NADPH depleted → glutathione can’t be reduced → hemoglobin is oxidized → hemoglobin denatures and precipitates → forms Heinz bodies (intracellular inclusions) → spleen tries to remove Heinz bodies → pulls out chunks of cytoplasm → bite cells (eccentrocytes).
Classic Triggers for G6PD Hemolytic Crisis:
- Antimalarials: Primaquine, chloroquine
- Antibiotics: Dapsone, sulfonamides, nitrofurantoin
- Infections (most common trigger overall)
- Fava beans (favism)
- Vitamin C in large doses
G6PD Epidemiology: X-linked recessive — most common in African American men, Mediterranean populations, and Southeast Asians. Heterozygous women are protected from severe malaria (selective advantage).
Lab findings in hemolytic anemia (any cause):
- ↓ Hemoglobin (and hematocrit)
- ↑ LDH (released from lysed RBCs)
- ↑ Indirect bilirubin (from heme catabolism)
- ↓ Haptoglobin (binds free hemoglobin → saturated and cleared)
- Hemoglobinuria → dark urine
Why the distractors are wrong:
- A (Autoimmune/IgG): That’s warm autoimmune hemolytic anemia (AIHA) — DAT (Coombs test) positive; no drug trigger pattern like this
- B (Spectrin deficiency): That’s Hereditary Spherocytosis — spherocytes on smear, NOT bite cells; osmotic fragility test positive
- C (Iron deficiency): Causes microcytic, hypochromic anemia — not hemolytic; no elevated LDH or bilirubin acutely
- E (Mechanical/fibrin strands): That’s Microangiopathic hemolytic anemia (MAHA) — seen in TTP, HUS, DIC; smear shows schistocytes (fragmented RBCs), not bite cells
🔴 QUESTION 7 — Organ Pathology: Renal (High-Yield)
A 7-year-old boy presents with periorbital edema, frothy urine, and weight gain for the past 2 weeks. Labs show: serum albumin 1.8 g/dL, total protein 4.2 g/dL, urinalysis with 4+ protein, no RBCs or casts. 24-hour urine protein is 4.8 g/day. Renal biopsy shows normal glomeruli on light microscopy, negative immunofluorescence, and effacement of podocyte foot processes on electron microscopy. What is the most likely diagnosis?
A) Focal segmental glomerulosclerosis (FSGS)
B) Membranous nephropathy
C) Minimal change disease
D) IgA nephropathy
E) Post-streptococcal glomerulonephritis
✅ Correct Answer: C — Minimal change disease
Detailed Explanation:
This is a nephrotic syndrome question, and the NBME tests glomerular diseases relentlessly because they force you to integrate clinical presentation, lab findings, and pathological findings simultaneously.
First: Identify the Syndrome — Nephrotic vs. Nephritic:
| Feature | Nephrotic | Nephritic |
|---|---|---|
| Proteinuria | Massive (>3.5 g/day) | Mild to moderate |
| Hematuria | Rare | Classic (RBCs, RBC casts) |
| Edema | Severe (periorbital, peripheral, ascites) | Mild |
| Hypertension | Variable | Common |
| Mechanism | Podocyte/GBM injury → protein leak | Inflammatory → GBM destruction |
This patient has massive proteinuria, hypoalbuminemia, edema → nephrotic syndrome.
Now identify the specific disease using the biopsy:
- Light microscopy: Normal → immediately narrows to Minimal Change Disease (MCD)
- Immunofluorescence: Negative → rules out immune complex diseases (membranous, MPGN, lupus nephritis, post-strep GN, IgA)
- Electron microscopy: Podocyte foot process effacement → the pathological hallmark of MCD
Minimal Change Disease Pearls:
- Most common cause of nephrotic syndrome in children
- Associated with Hodgkin lymphoma in adults (cytokines from Reed-Sternberg cells damage podocytes) and NSAIDs
- Responds dramatically to corticosteroids (excellent prognosis in children)
- Pathogenesis: T cell dysfunction → cytokines damage podocytes → podocyte foot process effacement → loss of negative charge on GBM → massive proteinuria
Glomerular Disease Quick Reference:
| Disease | Syndrome | LM | IF | EM | Key Association |
|---|---|---|---|---|---|
| Minimal Change Disease | Nephrotic | Normal | Negative | Foot process effacement | Children, Hodgkin lymphoma |
| Focal Segmental GS | Nephrotic | Focal/segmental sclerosis | IgM, C3 | Foot process effacement | HIV, heroin, obesity, sickle cell |
| Membranous Nephropathy | Nephrotic | Thickened GBM, “spike and dome” | IgG, C3 granular | Subepithelial deposits | Adults, #1 in adults; HBV, SLE |
| IgA Nephropathy | Nephritic | Mesangial hypercellularity | IgA deposits | Mesangial deposits | Young male, follows URI |
| Post-strep GN | Nephritic | “Lumpy bumpy” hypercellularity | IgG, C3 granular | Subepithelial “humps” | Children, follows strep infection |
| Goodpasture Syndrome | Nephritic | Crescents | IgG linear | Subendothelial deposits | Anti-GBM antibodies, lung hemorrhage |
High-Yield Pathology Concepts You Must Master for Step 1
Cell Death: Apoptosis vs. Necrosis
Students confuse these. The NBME tests them constantly.
Necrosis is passive, caused by external injury (ischemia, toxins, infection). It causes cell swelling, membrane rupture, and inflammation — the contents spill out and damage neighboring cells.
Apoptosis is programmed, physiological or pathological. It is characterized by cell shrinkage, chromatin condensation (pyknosis), nuclear fragmentation (karyorrhexis), membrane blebbing, and apoptotic bodies — which are phagocytosed cleanly. No inflammation.
Two apoptosis pathways:
- Intrinsic (mitochondrial): DNA damage, hypoxia → cytochrome c release → caspase activation. Regulated by BCL-2 family (BCL-2 inhibits apoptosis; BAX promotes it). BCL-2 overexpression = lymphoma (follicular lymphoma, t[14;18])
- Extrinsic (death receptor): FasL binds Fas (CD95) → caspase-8 activation → execution caspases → cell death
Liquefactive necrosis in the brain: Neurons are rich in phospholipids and the brain has abundant hydrolytic enzymes → tissue liquefies instead of coagulating.
Wound Healing: Primary vs. Secondary Intention
Primary intention: Clean surgical incision, edges approximated → epithelial cells migrate across within 24–48 hours → collagen deposition peaks at 6–8 weeks → scar.
Secondary intention: Large open wound → granulation tissue fills defect → wound contraction (myofibroblasts) → scar. More prominent scar, longer healing time.
Collagen timeline (USMLE favorite):
- Day 1–2: Clot, inflammation, neutrophils
- Day 3–5: Macrophages (“cleaning crew”), early granulation tissue, angiogenesis
- Week 1: Fibroblasts deposit Type III collagen (first collagen in wound)
- Week 4+: Type I collagen replaces Type III (stronger)
- Tensile strength never fully recovers — maximum ~80% of normal
Complications of wound healing:
- Dehiscence: Wound reopens (Vitamin C deficiency → poor collagen synthesis)
- Keloid: Scar grows beyond original wound margins. More common in African Americans; hypertrophic scar stays within wound margins.
- Proud flesh: Excessive granulation tissue that prevents epithelialization
Tumor Grading vs. Staging — A Distinction the NBME Loves
Grading = pathological assessment of how differentiated the tumor cells appear under the microscope. It reflects biological behavior.
- Low grade = well-differentiated = slower growing
- High grade = poorly differentiated/anaplastic = aggressive
Staging = clinical/pathological extent of tumor spread. Uses TNM system (Tumor size, Node involvement, Metastasis). It reflects how far the cancer has spread.
“Grade is the microscope; Stage is the map.”
Which matters more for prognosis? Generally, staging has more clinical prognostic weight. However, for specific tumors (like bladder cancer, prostate cancer), grading is especially important.
The Five Key Tumor Markers (USMLE High-Yield)
| Marker | Associated Tumors |
|---|---|
| AFP (α-fetoprotein) | Hepatocellular carcinoma, yolk sac tumor (testicular), neural tube defects (elevated in amniotic fluid) |
| β-hCG | Choriocarcinoma, gestational trophoblastic disease, some germ cell tumors |
| CEA (carcinoembryonic antigen) | Colorectal cancer, pancreatic, gastric, lung — NOT specific, used for monitoring recurrence |
| PSA | Prostate cancer (also elevated in BPH, prostatitis — NOT perfectly specific) |
| CA-125 | Ovarian cancer (serous carcinoma) — used for monitoring |
| CA 19-9 | Pancreatic cancer |
| S-100 | Melanoma, Schwannoma, Langerhans cell histiocytosis |
| TRAP (tartrate-resistant acid phosphatase) | Hairy cell leukemia |
Study Strategy: How to Dominate Pathology on Step 1
1. Build “Disease Profiles” Not Fact Lists
For every major disease, build a mental template:
Mechanism → Morphology (gross + histological findings) → Clinical Presentation → Complications → Treatment
When you learn a disease this way, any USMLE question — regardless of which angle it approaches from — feeds into the same mental framework.
2. Use Pathoma as Your Foundation
Dr. Hussain Sattar’s Pathoma (Fundamentals of Pathology) is the gold standard pathology resource for Step 1. Its chapters align perfectly with organ systems, and his teaching style emphasizes mechanism over memorization. Read it alongside First Aid.
3. Do Questions from Day One (Not After “Finishing” the Material)
One of the biggest mistakes Step 1 students make is waiting to do questions until they’ve “finished studying.” Start UWorld pathology questions as early as week 2 of dedicated study. You don’t need to know everything — questions teach you what you don’t know and how to think.
4. Master Histology Through Images, Not Words
Pathology is visual. When reviewing a disease, always pair your text reading with the corresponding histology image. Amboss has exceptional histology images integrated with Q-bank questions. Anki decks with histology images (like Anki’s Pathoma deck) are invaluable.
5. Create a “Buzzwords → Disease” Cheat Sheet
Many Step 1 pathology questions drop specific buzzwords. Build a running list:
| Buzzword | Disease |
|---|---|
| “Apple-green birefringence under polarized light” | Amyloid (Congo red stain) |
| “Onion skin” periosteal reaction | Ewing sarcoma |
| “Codman triangle” on X-ray | Osteosarcoma |
| “Boot-shaped heart” on CXR | Tetralogy of Fallot (or right ventricular hypertrophy) |
| “Cafe-au-lait spots” + neurofibromas | NF-1 (Neurofibromatosis type 1) |
| “Auer rods” in blasts | AML (Acute Myeloid Leukemia) |
| “Reed-Sternberg cells” (owl eye) | Hodgkin Lymphoma |
| “Keratin pearls” in epithelium | Squamous Cell Carcinoma |
| “Psammoma bodies” | Papillary thyroid carcinoma, meningioma, serous ovarian carcinoma |
| “Call-Exner bodies” | Granulosa cell tumor (ovary) |
| “Reinke crystals” | Leydig cell tumor (testis) |
| “Signet ring cells” | Gastric adenocarcinoma (diffuse type) |
| “Druse bodies” / geographic atrophy | Macular degeneration |
Quick Recall Quiz — Test Yourself
Try these before scrolling to answers:
- What type of necrosis occurs in a brain abscess?
- A 40-year-old woman with a “butterfly rash” and positive ANA — what type of hypersensitivity is predominantly responsible for her renal disease?
- Which tumor suppressor is mutated in both retinoblastoma and osteosarcoma?
- What is the most common cause of death in patients with SLE?
- A peripheral blood smear shows hypersegmented neutrophils and macro-ovalocytes. What deficiency is most likely?
Answers:
- Liquefactive necrosis (pus = liquefactive; brain also = liquefactive)
- Type III (immune complex deposition — SLE causes immune complex GN via antigen-antibody deposits in glomeruli)
- RB1 (retinoblastoma gene — classic two-hit hypothesis; retinoblastoma in eye, osteosarcoma in long bones)
- Renal failure / lupus nephritis (most common cause of death in SLE is renal disease)
- Vitamin B12 or folate deficiency → megaloblastic anemia with impaired DNA synthesis in rapidly dividing cells
usmle biostatistics practice questions step 1
Conclusion: From Passive Reader to Active Pathologist
Pathology is not about memorizing more facts than the next student. It’s about building a mental model of how disease works — at the cellular level, the tissue level, and the whole-patient level. The NBME writes questions assuming you can move fluently between these levels.
Every practice question you do correctly reinforces a pathway. Every wrong answer — if you study the explanation deeply — teaches you something that will come back on exam day. The best pathology students treat every incorrect answer not as a failure, but as a gift: a preview of exam content they now own.
Work through real USMLE-style questions, understand every mechanism, build your buzzword list, and trust the process. Step 1 is not about who studies the most hours — it’s about who studies the most intelligently.
Disclaimer: This content is for educational purposes related to USMLE Step 1 examination preparation only. It does not constitute medical advice. Always consult licensed medical professionals for clinical decisions.
